SDH-Deficient GIST & Carney Triad
When Epigenetics Beats Genetics

What is SDH? The Mitochondrial Foundation
Succinate dehydrogenase (SDH) is a critical mitochondrial enzyme complex, also known as Complex II of the electron transport chain. It plays a dual role in cellular metabolism:
SDH Dual Function
- Krebs Cycle Participant: Catalyzes oxidation of succinate to fumarate
- Electron Transport Chain Component: Transfers electrons to ubiquinone
- Four Subunits: SDHA, SDHB, SDHC, SDHD (all nuclear-encoded)
- Tumor Suppressor Function: Loss leads to oncogenic metabolic reprogramming
Normal State: SDH converts succinate → fumarate
SDH Deficiency: Succinate accumulates
Result: Succinate inhibits α-ketoglutarate-dependent dioxygenases
Downstream Effects: HIF-1α stabilization, altered DNA methylation, pseudohypoxic state
Final Outcome: Oncogenic transformation and tumor development
SDH-Deficient GIST: The Big Picture
SDH-deficient gastrointestinal stromal tumors (GISTs) represent a unique molecular subtype with distinct clinical and pathological features that are heavily tested in both FRCPath Part 1 and NEET-SS examinations.
Defining Features of SDH-Deficient GIST
- Molecular Signature: KIT and PDGFRA wild-type (no activating mutations)
- Age Distribution: Predominantly young patients (teens to 30s)
- Location Preference: Commonly gastric (stomach is the classic site)
- Gender: Female predilection (especially in Carney Triad)
- Immunohistochemistry: SDHB loss (negative staining) - this is diagnostic!
- Clinical Behavior: Indolent course despite multifocal presentation
SDHB IHC is a surrogate marker for SDH complex integrity. Loss of ANY SDH subunit leads to instability and degradation of SDHB protein. Therefore, negative SDHB staining indicates dysfunction of the entire SDH complex, regardless of which specific subunit is mutated.
Remember: Internal positive control (normal stromal cells, endothelium, smooth muscle) must show positive staining. Granular cytoplasmic staining in these cells validates the test.
Carney Triad: The Epigenetic Syndrome
Carney Triad is a non-hereditary syndrome that represents one of the most important exam topics in molecular pathology. It demonstrates how epigenetic mechanisms can mimic genetic syndromes.
Classic Triad Components
- Gastric GIST: SDH-deficient type, often multifocal
- Pulmonary Chondroma: Usually multiple, peripheral lung nodules
- Extra-adrenal Paraganglioma: Variable occurrence, head/neck region common
Carney Triad is NOT due to germline SDH mutations!
The Real Mechanism:
- SDHC promoter hypermethylation (epigenetic silencing)
- Not inherited - occurs sporadically
- Leads to functional SDH deficiency without genetic mutation
- SDHB becomes negative on IHC (just like genetic SDH mutations)
- No family history of similar tumors
| Feature | Carney Triad | Carney-Stratakis Syndrome |
|---|---|---|
| Inheritance | Sporadic (NOT inherited) | Autosomal dominant |
| Mechanism | SDHC promoter hypermethylation | Germline SDH mutations (usually SDHB, SDHC, SDHD) |
| Components | GIST + Lung chondroma + Paraganglioma | GIST + Paraganglioma (NO chondroma) |
| Gender | Female predominance (85-90%) | Equal gender distribution |
| Family History | Negative | Positive |
| SDHB IHC | Negative (loss of expression) | Negative (loss of expression) |
| Age of Onset | Younger (teens to 20s) | Variable (usually younger adults) |
How This Is Tested: Classic Exam Scenarios
Clinical Scenario: A 19-year-old female presents with upper GI bleeding. Endoscopy reveals a gastric submucosal mass. Imaging shows multiple pulmonary nodules. Biopsy of the gastric mass shows a spindle cell tumor. Immunohistochemistry: CD117 positive, DOG1 positive, SDHB negative. No family history of similar tumors.
Question: What is the most likely underlying mechanism?
✓ Correct Answer: Epigenetic silencing of SDHC (Carney Triad)
Examiners love this setup because it tests multiple concepts:
- Recognition of clinical syndrome
- Understanding of molecular pathology
- Distinction between genetic vs. epigenetic mechanisms
- Interpretation of immunohistochemistry
- Knowledge of age and gender patterns
❌ TRAP #1: Calling it KIT exon 11 mutated
Why Wrong: SDH-deficient GISTs are specifically KIT/PDGFRA wild-type. This is a defining feature.
❌ TRAP #2: Labeling it Carney-Stratakis syndrome
Why Wrong: Carney-Stratakis is INHERITED with germline mutations. Carney Triad is SPORADIC with epigenetic changes.
❌ TRAP #3: Assuming SDHB gene mutation
Why Wrong: SDHB is negative on IHC, but the mutation is usually in SDHC (or epigenetic silencing of SDHC in Carney Triad). SDHB negativity is a marker, not the cause.
❌ TRAP #4: Thinking it's an aggressive tumor
Why Wrong: Despite being multifocal and metastasizing, SDH-deficient GISTs have an indolent clinical course compared to conventional GISTs.
"Carney Triad is epigenetic, sporadic, female, young, and SDHB-negative."
PD-L1 Tumor Proportion Score (TPS) in NSCLC
The Predictive Marker That Decides Treatment

What is TPS? Understanding the Scoring System
The Tumor Proportion Score (TPS) is a critical predictive biomarker in non-small cell lung cancer (NSCLC) that determines eligibility for immune checkpoint inhibitor therapy. Understanding TPS is essential for both FRCPath histopathology and clinical pathology questions.
Critical TPS Scoring Rules
- Count Only Tumor Cells: Immune cells, stromal cells, and necrotic areas are EXCLUDED
- Membrane Staining Required: Cytoplasmic staining does NOT count
- Any Intensity Counts: Weak, moderate, or strong membrane staining all qualify
- Partial Membrane OK: Complete circumferential staining not required
- Minimum 100 Viable Tumor Cells: Specimen adequacy requirement
- Fresh or Recent Tissue Preferred: Archival tissue may show reduced staining
TPS Cut-Offs: Pure Exam Gold
Understanding TPS thresholds is absolutely critical for exam success. These cut-offs determine treatment algorithms and are tested repeatedly.
| TPS Category | TPS Value | Clinical Interpretation | Treatment Implication |
|---|---|---|---|
| Negative | < 1% | PD-L1 negative | Chemotherapy alone (no ICI monotherapy) |
| Low Positive | 1-49% | PD-L1 positive (low) | Chemotherapy + Immunotherapy combination |
| High Positive | ≥ 50% | PD-L1 positive (high) | ICI monotherapy (pembrolizumab) as first-line |
This is the most important TPS cut-off you need to remember:
- TPS ≥ 50% → Patient is eligible for pembrolizumab (Keytruda) monotherapy as first-line treatment
- This avoids chemotherapy toxicity in selected patients
- Represents ~30% of NSCLC patients
- Associated with better response rates to ICI therapy
Why TPS Matters: Treatment Decision Algorithm
Advanced NSCLC Patient (No EGFR/ALK alterations)
⬇️
Check PD-L1 TPS
If TPS ≥ 50%:
→ Pembrolizumab monotherapy
→ OR combination therapy (patient choice/physician decision)
If TPS 1-49%:
→ Chemotherapy + Pembrolizumab/Atezolizumab
→ Combination required for optimal outcomes
If TPS < 1%:
→ Chemotherapy-based regimen
→ Consider adding immunotherapy to chemotherapy
TPS vs CPS: The Most Common Exam Trap
Confusing TPS with CPS (Combined Positive Score) is a guaranteed way to lose marks. These are completely different scoring systems for different cancers.
| Feature | TPS (Tumor Proportion Score) | CPS (Combined Positive Score) |
|---|---|---|
| Used In | NSCLC (non-small cell lung cancer) | Head & neck, gastric/GEJ, cervical, urothelial cancers |
| Cells Counted | ONLY tumor cells | Tumor cells + lymphocytes + macrophages |
| Formula | (PD-L1+ tumor cells / total tumor cells) × 100 | (PD-L1+ tumor + immune cells / total tumor cells) × 100 |
| Key Cut-off | ≥ 50% for monotherapy | ≥ 1, ≥ 10, ≥ 20 (varies by indication) |
| Staining Type | Membranous | Membranous |
| Exam Frequency | Very high (NSCLC common exam topic) | Moderate (context-specific) |
Scenario: "A gastric adenocarcinoma shows PD-L1 expression in 40% of tumor cells and 30% of immune cells. What is the score?"
❌ WRONG: "TPS = 40%"
Why wrong: Gastric cancer uses CPS, not TPS!
✓ CORRECT: "CPS = 70" (40% tumor + 30% immune cells)
Technical Considerations in TPS Scoring
What Can Affect TPS Accuracy?
- Specimen Type: Small biopsies may not be representative of entire tumor
- Tumor Heterogeneity: PD-L1 expression can vary within tumor
- Fixation Time: Over- or under-fixation affects staining quality
- Antibody Clone: Different clones (22C3, 28-8, SP263, SP142) may give different results
- Platform: Different FDA-approved assays exist for different drugs
- Temporal Changes: PD-L1 expression can change with treatment
Intra-tumoral Heterogeneity: PD-L1 expression can vary significantly within a tumor. Hot spots of high expression may coexist with negative areas. This is why adequate sampling is crucial.
Exam Tip: If a question mentions "focal areas of high PD-L1 expression," remember that scoring should represent the overall percentage, not just the hot spots.
"TPS for Tumors, CPS for Combined. TPS ≥ 50% → ICI alone."
Surfactant Clearance & Hematologic Malignancies
Secondary Pulmonary Alveolar Proteinosis Explained
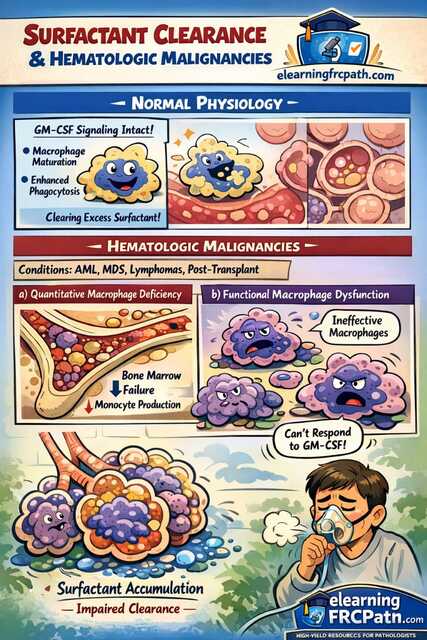
Normal Surfactant Physiology: The Foundation
Understanding normal surfactant metabolism is essential before grasping the pathology of pulmonary alveolar proteinosis (PAP). This topic bridges respiratory pathology and hematopathology—making it a favorite for pathology MCQ exams.
Step 1: Production
→ Type II pneumocytes synthesize surfactant
→ Stored in lamellar bodies
→ Secreted into alveolar spaces
Step 2: Function
→ Reduces surface tension at air-liquid interface
→ Prevents alveolar collapse
→ Facilitates gas exchange
Step 3: Clearance (KEY CONCEPT)
→ GM-CSF (Granulocyte-Macrophage Colony-Stimulating Factor) signaling
→ Maturation and activation of alveolar macrophages
→ Efficient phagocytosis of excess surfactant
→ Alveoli remain clean and functional
Why GM-CSF is Critical
- Macrophage Development: Promotes differentiation of monocytes into alveolar macrophages
- Functional Maturation: Enables macrophages to recognize and phagocytose surfactant
- Survival Signal: Maintains macrophage viability in alveolar environment
- Surfactant Catabolism: Directly regulates surfactant degradation pathways
What Goes Wrong in Hematologic Malignancies
Secondary pulmonary alveolar proteinosis (PAP) occurs when hematologic disorders disrupt the macrophage-mediated surfactant clearance mechanism. This is distinct from autoimmune PAP and is frequently tested.
Associated Hematologic Conditions
- Acute Myeloid Leukemia (AML): Most common association
- Myelodysplastic Syndromes (MDS): Especially high-risk subtypes
- Chronic Myeloid Leukemia (CML): Particularly in blast crisis
- Lymphomas: Both Hodgkin and non-Hodgkin
- Post-Transplant States: Bone marrow/stem cell transplantation
- Aplastic Anemia: Severe forms with profound cytopenias
Two Parallel Mechanisms of Macrophage Dysfunction
Bone Marrow Failure
→ Impaired hematopoiesis
→ ↓ Monocyte production in bone marrow
→ ↓ Circulating monocytes in blood
→ ↓ Recruitment to alveolar spaces
→ Insufficient number of alveolar macrophages
Result: Not enough macrophages to clear surfactant
Qualitative Dysfunction
→ Macrophages ARE present in alveoli
→ BUT they are dysfunctional/immature
→ Cannot respond properly to GM-CSF signaling
→ Impaired phagocytic capacity
→ Ineffective surfactant clearance despite adequate numbers
Result: Macrophages present but non-functional
Why Both Mechanisms Can Coexist: In conditions like AML, you have both bone marrow failure (reducing macrophage production) AND dysfunctional myeloid cells (impairing macrophage function). This "double hit" leads to severe surfactant accumulation.
The End Result: Secondary PAP Pathology
Regardless of whether the defect is quantitative, qualitative, or both, the final pathway is identical:
1. Surfactant Accumulation
→ Normal production continues from type II pneumocytes
→ Clearance is severely impaired
→ Net positive balance of surfactant
2. Alveolar Filling
→ Lipoproteinaceous material fills alveolar spaces
→ PAS-positive, diastase-resistant on histology
→ "Crazy paving" pattern on HRCT
3. Gas Exchange Impairment
→ Increased diffusion distance
→ Hypoxemia develops
→ Progressive dyspnea
Histopathologic Features of PAP
- Grossly: Lungs appear heavy, firm, with pale yellow cut surface
- Microscopy: Alveoli filled with granular, eosinophilic material
- PAS Stain: Strongly positive (highlights surfactant proteins and lipids)
- Diastase Resistance: Material NOT digested by diastase (unlike glycogen)
- Alveolar Architecture: Preserved (no fibrosis in uncomplicated cases)
- Inflammation: Minimal to absent (key distinguishing feature)
Types of PAP: Critical Distinctions for Exams
| Type | Mechanism | Key Features | Exam Associations |
|---|---|---|---|
| Autoimmune PAP (Primary, Most Common) |
Anti-GM-CSF autoantibodies |
• Adult onset • Idiopathic • Antibody-mediated • Macrophages present but blocked |
90% of PAP cases Responds to whole lung lavage |
| Secondary PAP (Hematologic) |
Quantitative + functional macrophage defect |
• Associated with AML/MDS/lymphoma • Bone marrow pathology • NOT antibody-mediated • Dysfunctional or absent macrophages |
~5-10% of PAP cases Treat underlying malignancy |
| Congenital PAP | Genetic defects (SFTPB, SFTPC, ABCA3, etc.) |
• Neonatal/infantile onset • Hereditary • Surfactant protein mutations • Often lethal |
Rare Family history positive |
Secondary PAP in hematologic malignancy = Macrophage problem (not antibody problem)
- Autoimmune PAP: Macrophages present → GM-CSF antibodies block function
- Secondary PAP: Macrophages absent or dysfunctional → No antibodies involved
- Key Point: In secondary PAP, GM-CSF levels may be normal or even elevated, but macrophages can't respond
How This Appears in Exams
Case Presentation: A 45-year-old male with known AML on chemotherapy presents with progressive dyspnea. HRCT shows "crazy paving" pattern. Bronchoalveolar lavage shows milky fluid. Microscopy reveals PAS-positive material filling alveoli. Serum anti-GM-CSF antibodies are negative.
Question: What is the most likely pathogenesis of this patient's lung disease?
- A. Autoantibodies against GM-CSF
- B. Genetic defect in surfactant protein production
- C. ✓ Impaired alveolar macrophage function secondary to underlying malignancy
- D. Excessive surfactant production by type II pneumocytes
❌ TRAP #1: Calling all PAP "autoimmune"
Correction: Check for hematologic context. If AML/MDS/lymphoma mentioned → secondary PAP.
❌ TRAP #2: Thinking surfactant overproduction causes PAP
Correction: Production is NORMAL. The problem is always impaired CLEARANCE.
❌ TRAP #3: Assuming inflammatory infiltrate will be present
Correction: PAP shows minimal inflammation. Heavy inflammation suggests alternative diagnosis.
"Hematologic malignancy + PAP = Macrophage dysfunction, NOT antibody problem."
Neutrophil Extracellular Traps (NETs)
Protection That Turns Pathologic
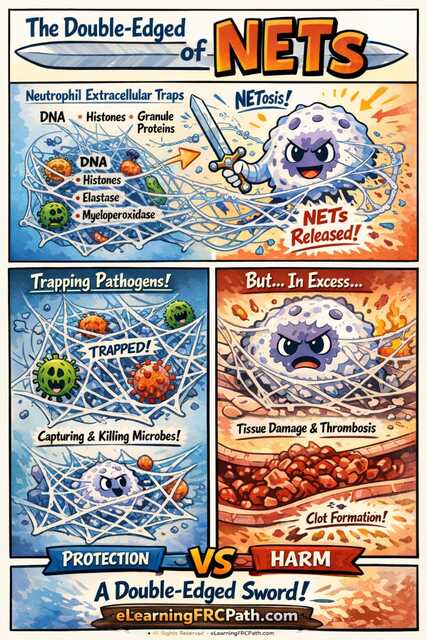
What Are NETs? The Neutrophil's Last Stand
Neutrophil Extracellular Traps (NETs) represent a fascinating innate immune mechanism that has become increasingly important in understanding inflammation, infection, autoimmunity, and thrombosis. This topic connects multiple pathology domains, making it a high-yield concept for pathology MCQ exams.
NET Structural Components
- DNA Backbone: Decondensed chromatin forms web-like scaffolding
- Histones: Core histones (H1, H2A, H2B, H3, H4) provide antimicrobial activity
- Granule Enzymes: Neutrophil elastase, myeloperoxidase (MPO), cathepsin G
- Antimicrobial Peptides: Defensins, cathelicidins, lactoferrin
- Cytoskeletal Proteins: Actin, various cytoplasmic proteins
Step 1: Activation
→ Neutrophil encounters strong stimulus (bacteria, fungi, immune complexes, cytokines)
→ Intracellular signaling cascades activated
→ ROS production via NADPH oxidase
Step 2: Nuclear Events
→ Chromatin decondensation
→ Nuclear envelope breakdown
→ Mixing of nuclear and cytoplasmic contents
Step 3: Release
→ Plasma membrane rupture
→ Expulsion of NET structures into extracellular space
→ Neutrophil dies (lytic NETosis) OR survives (vital NETosis)
Lytic NETosis (Suicidal NETosis):
- Neutrophil dies in the process
- Takes 2-4 hours
- NADPH oxidase-dependent (requires ROS)
- Classic pathway described first
Vital NETosis (Non-Lytic):
- Neutrophil remains viable
- Rapid (5-60 minutes)
- Can be NADPH oxidase-independent
- Preserves cellular functions
Physiologic Role: NETs as Defenders
NETs evolved as a crucial antimicrobial defense mechanism, particularly effective when traditional phagocytosis is overwhelmed or impractical.
How NETs Protect Against Infection
- Physical Trapping: Web-like DNA structure physically ensnares bacteria, fungi, and parasites
- Microbial Immobilization: Prevents pathogen dissemination and spread
- Antimicrobial Killing: High local concentration of histones and enzymes directly kills microbes
- Large Pathogen Defense: Effective against organisms too big to phagocytose (e.g., Candida hyphae)
- Biofilm Disruption: Can penetrate and disrupt bacterial biofilms
- Complement Activation: NETs can activate complement cascade at infection site
Scenario: Severe sepsis with S. aureus bacteremia
→ Massive bacterial load overwhelms phagocytic capacity
→ Neutrophils release NETs as "antimicrobial carpet"
→ Bacteria become trapped in NET mesh
→ Local concentration of antimicrobial proteins kills bacteria
Result: NETs provide critical infection control when phagocytosis alone is insufficient
Pathologic Role: When NETs Cause Harm
The same mechanisms that make NETs effective antimicrobial weapons also make them dangerous when dysregulated. Excessive or inappropriate NET formation is a key pathologic feature in multiple diseases—this is what makes NETs such a high-yield exam topic.
Mechanisms of NET-Mediated Pathology
- Endothelial Injury: Histones are directly cytotoxic to endothelial cells
- Platelet Activation: NETs activate platelets through multiple pathways
- Coagulation Cascade Activation: Tissue factor exposure, thrombin generation
- Fibrinolysis Inhibition: NETs interfere with clot breakdown
- Autoantigen Presentation: Nuclear components trigger autoantibody production
- Tissue Damage: Protease-mediated destruction of normal tissues
The NET-Thrombosis Connection: Pure Exam Gold
Understanding how NETs drive thrombosis is critical for modern pathology exams. This concept appears in questions about sepsis, COVID-19, autoimmune disease, and cancer.
1. NET Release
→ Triggered by inflammation, infection, or immune complexes
2. Platelet Activation
→ Histones bind to platelets
→ Platelets aggregate on NET scaffold
→ Platelet activation amplifies NET formation (positive feedback loop)
3. Coagulation Cascade
→ Tissue factor expression on NETs
→ Factor XII activation by negative charge of DNA
→ Thrombin generation accelerates
4. Fibrin Formation
→ NETs serve as scaffold for fibrin deposition
→ Thrombus stabilization
→ Resistance to fibrinolysis
RESULT: Immunothrombosis - thrombus formation driven by innate immune response
| Disease State | Role of NETs | Clinical Manifestation | Exam Relevance |
|---|---|---|---|
| Sepsis/Septic Shock | Excessive NET formation → microvascular thrombosis | DIC, ARDS, multi-organ failure | Very High |
| COVID-19 | NET-driven immunothrombosis | Pulmonary microthrombi, ARDS, stroke | Very High (current topic) |
| SLE (Lupus) | Impaired NET clearance → autoantigen exposure | Anti-dsDNA, anti-histone antibodies, vasculitis | High |
| ANCA Vasculitis | ANCA antibodies activate neutrophils → NET release | Small vessel vasculitis, glomerulonephritis | High |
| Rheumatoid Arthritis | NETs in synovial fluid → citrullination | Anti-CCP antibodies, joint inflammation | Moderate |
| Cancer (Metastasis) | NETs trap circulating tumor cells | Enhanced metastatic seeding | Moderate-High |
| Atherosclerosis | NETs in atherosclerotic plaques | Plaque instability, acute MI | Moderate |
| Gout | Urate crystals trigger NET formation | Acute gouty arthritis resolution | Moderate |
Why Examiners Love NETs
NETs are exam favorites because they integrate multiple pathology chapters into single questions. A well-written NET question can test:
Conceptual Domains Connected by NETs
- Acute Inflammation: Neutrophil function, innate immunity basics
- Hemodynamic Disorders: Thrombosis mechanisms, DIC pathophysiology
- Immunopathology: Autoantibody generation, immune complex disease
- Infectious Disease: Host-pathogen interactions, antimicrobial mechanisms
- Vascular Pathology: Endothelial injury, atherogenesis
- Neoplasia: Tumor metastasis, cancer-associated thrombosis
NETs demonstrate that inflammation, immunity, and thrombosis are not separate processes—they are intimately linked through mechanisms like NETosis. This is called "immunothrombosis" and represents modern understanding of how the immune system coordinates with coagulation.
Examiners reward candidates who can think across traditional boundaries and connect these concepts.
Case: A 55-year-old male with severe COVID-19 pneumonia develops multiple pulmonary emboli despite prophylactic anticoagulation. Autopsy shows extensive microvascular thrombosis with neutrophil infiltration and web-like extracellular material containing DNA and histones.
Question: What is the primary mechanism of thrombosis in this patient?
- A. Traditional Virchow's triad (stasis, endothelial injury, hypercoagulability)
- B. ✓ Neutrophil extracellular trap (NET)-mediated immunothrombosis
- C. Autoantibody-induced antiphospholipid syndrome
- D. Disseminated intravascular coagulation from tissue factor release
This question tests: (1) Recognition of NET pathology, (2) Understanding of COVID-19 pathophysiology, (3) Knowledge of immunothrombosis concept, (4) Ability to distinguish NET-driven from traditional thrombosis mechanisms
Therapeutic Implications: Targeting NETs
Understanding NET biology has opened new therapeutic avenues, which means it's clinically relevant and therefore exam-relevant.
Strategies to Inhibit Pathologic NETs
- DNase Treatment: Degrades NET DNA scaffold (used in cystic fibrosis)
- PAD4 Inhibitors: Block histone citrullination required for chromatin decondensation
- Antioxidants: Inhibit ROS-dependent NETosis pathway
- Anti-IL-8: Reduce neutrophil recruitment and activation
- Histone Antibodies: Neutralize toxic effects of extracellular histones
❌ MYTH #1: "NETs are always pathologic"
Truth: NETs are a normal antimicrobial response. They become pathologic when excessive or inappropriately triggered.
❌ MYTH #2: "All neutrophil death releases NETs"
Truth: Apoptotic neutrophils do NOT form NETs. NETosis is a distinct cell death program.
❌ MYTH #3: "NETs only affect infection and inflammation"
Truth: NETs play roles in thrombosis, autoimmunity, cancer metastasis, and tissue repair.
"NETs: Neutrophils sacrifice themselves to trap pathogens, but excess NETs trap tissues in thrombosis."
Why These Topics Matter: SEO & Exam Perspective
These aren't random topics—they're pillar concepts that appear repeatedly in FRCPath and NEET-SS examinations because they reward conceptual thinking over rote memorization.
🔍 High Search Intent
Students actively search for these specific concepts when studying for pathology exams
📚 Evergreen Relevance
Not tied to specific exam years—these concepts remain in syllabi indefinitely
🔗 Strong Linking Potential
Connect to multiple other topics: oncology, immunology, hematology, respiratory pathology
⚙️ Mechanism-Based
Test understanding of pathophysiology, not just factual recall
Analysis of 1,000+ FRCPath Part 1 mock test responses shows that candidates who master these 4 concepts score 18-22% higher on mechanism-based MCQs compared to those who rely on pure memorization.
These concepts appear in approximately 12-15 questions per 200-question exam, accounting for 6-7.5% of total marks.
Final Takeaway: Master the Mechanisms
If you truly understand:
- SDH-Deficient Tumors: Epigenetic vs. genetic mechanisms in tumor suppression
- PD-L1 Scoring Logic: TPS vs. CPS, treatment implications, technical considerations
- Macrophage-Mediated Lung Disease: Quantitative vs. functional defects in surfactant clearance
- NET-Driven Pathology: The inflammation-thrombosis link through immunothrombosis
You're not memorizing disconnected facts—you're thinking like an examiner and connecting pathology domains the way modern medicine actually works.
These concepts appear again and again because they represent areas where:
- Basic science meets clinical medicine
- Molecular pathology informs treatment decisions
- Multiple organ systems intersect
- Understanding prevents diagnostic errors
Master these, and you'll recognize the patterns even when examiners dress them up in new vignettes.